The rate of evaporation, thus cooling is governed by the saturated vapour pressure value. It is a thermal dynamic topic, no chemical change has taken place.
Once the equilibrium is reached, no more cooling will take place untill temperature is increased or pressure reduced.
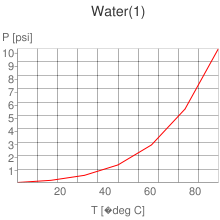
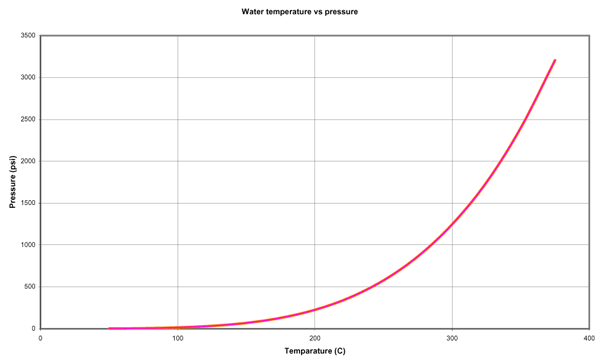
The following chart represents the total internal energy of water. At 370 C, water comes a gas (single molecule).
You can look this up on the steam table or enthalpy/entrogy property of water in any reference book.
Link to a simple calculation (cooling by water).
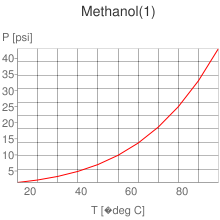
Substitude in the methanol properties to made a comparison.
http://www.aquamist.co.uk/rescr/faq/...rmass.html#cal